When a pandemic happens, there are many ways to write about it. You could write about the virus itself, the biological reasons these tiny protein particles spread so quickly and so dangerously. You could also write about how the way we get information online can lead to a wide misunderstanding of the issues. Both are key to this pandemic, and both need to be prevented.
So lets talk about both.
What does it mean to be ‘viral’?

We normally think of a viruses as tiny disease causing protein shells with the pointy bits on. The world’s deadliest pincushion or world’s angriest hedgehog. However, the word has come to take on another meaning in the modern day:
“Viral: something that is circulated rapidly and widely, particularly on the internet.”
Whilst there have been pandemics in the past, and less widespread ones such as Ebola and SARS since the internet first took hold of our lives, this is the first true pandemic of the digital age. We’re at a time when we all hold a direct line to the world in the palm of our hand. Mobile phone internet use accounts for half of ALL internet traffic. It has never been easier for so much of the world to access so much information, and yet it seems everyday we encounter people who aren’t in possession of all the facts, or actively believe falsehoods. Everyday we may be reading things ourselves that aren’t strictly true.
In a time of great fear and anxiety such as this there are those who seek to inform, and those who seek to misinform. There hasn’t been a more important time since the dawn of the internet to expose bogus therapies and medical misinformation, and there are many great blogs dedicated to debunking false claims and bad science (some I mention at the end). Some sites are even trying to cloud source fact checking to combat fake news on social media.
Yet however much effort we put in, fake facts always have an advantage. In a new topic the truth is constantly being learned, revised and updated, so truth-tellers will always be second place. It takes one word to ‘lie’, but two to ‘be truthful’.
Debunking every conspiracy theory isn’t my aim today. Partly because I didn’t think I’d require in depth knowledge of subjects like telephone communication networks to explain a viral disease.
We’re living in a situation with two parallel plagues, one of disease, and one of misinformation. They are inseparable in importance, and deeply linked with one another. You can use the same information to explain both how viruses spread through our bodies and how fake news spreads across the internet.
Misinformation may go viral, but biology was there first…
Part 1: Our cells and information
So first we’ll address the classic case of virality. The virus itself and how it spreads.
As with alot of things in biology, how a virus spread is down to its genetics. I talked about genes and their importance in a previous blog. They’ve been a part of us for billions of years, which if my perception of time has been right is about twice as long as we’ve been in lockdown for.
In that blog I describe our DNA, and the genes it makes up, as a microscopic instruction manual within every cell. These instructions are a guide for our cells to build, grow, replicate and thrive, and they’re many chapters long.
Which instructions are able to be read at what time is very carefully controlled by your cells. If we read an instruction manual alone, we simply follow the numbered steps like a recipe. But our cells have many different components, all of which need to work at the same time on different building blocks. It’s less like a recipe and more of its own little self-sufficient city. It has energy production (mitochondria), supply transport (golgi body and vesicles), stockpiles (vacuoles) and waste disposal (lysosomes).
The two most important bits for us today are the “command centre”, which we’ll think of a city hall, and “supply production” which is like a series of factories. Our cell’s command centre is known as the “nucleus”, and is where our DNA is stored, whilst the factories are “ribosomes” which are situated around the nucleus.

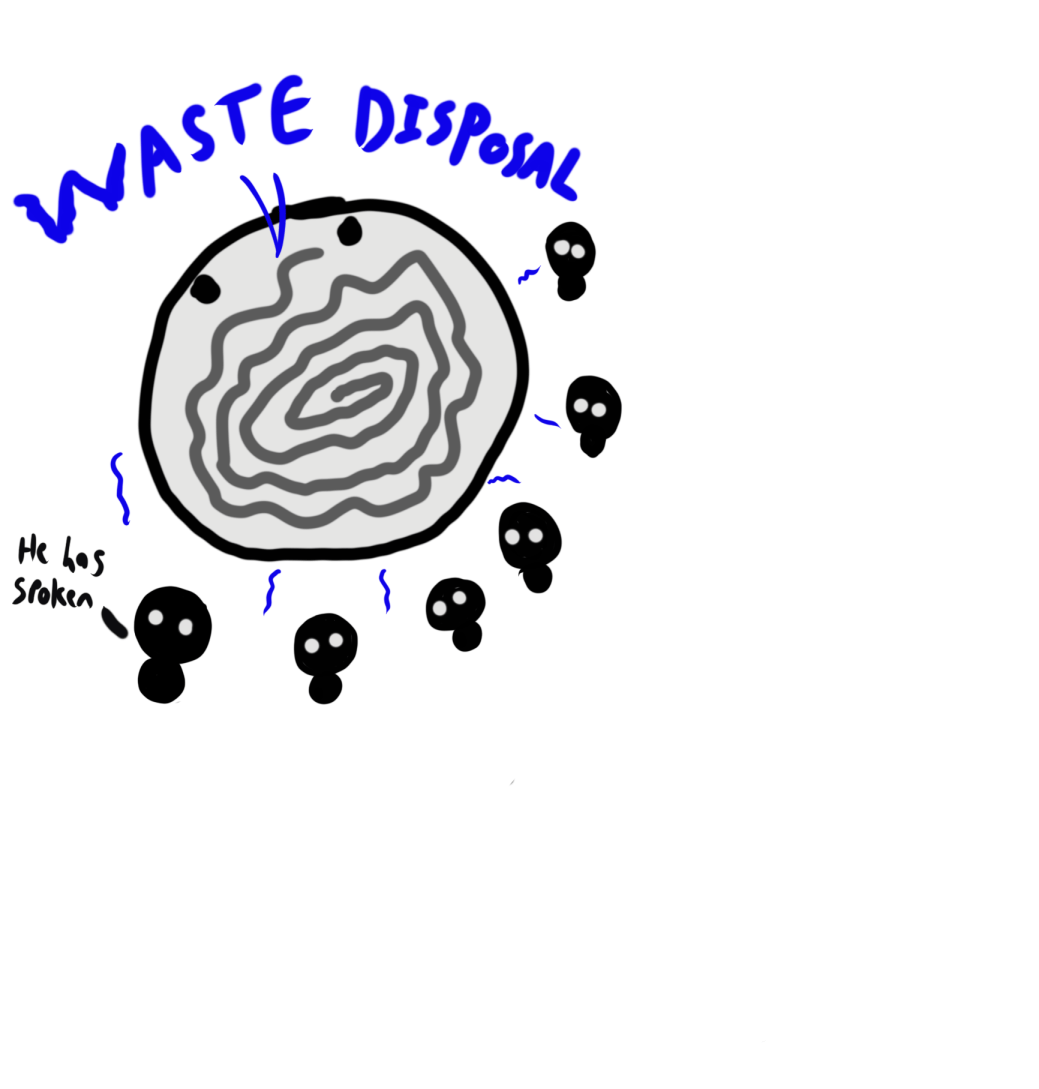
These factories can produce all the products required for the cell, but the cell doesn’t require all of these products all of the time. You don’t want to ramp up umbrella production at the start of summer, or start pumping out Christmas decorations in April. As a result, only small parts of the instructions, or “messages” are sent from the command centres to the factories, based on feedback about what is required. (These are the little blue lines in the image above)
In our example, our cell, or city, needs a new waste disposal site (exciting I know, but that’s town planning for you.) Our command centre at Cell City Hall will copy and send out the specific instructions on how to build a waste disposal site (It’s not just a hole in the ground, think more ’recycling centre’). These instructions then arrive at the factories.
Now, whilst the master copy of the instructions in the Cell City Hall is stored as DNA, this isn’t the best material for the small instructions sent out to the factories. DNA is very stable. I’m talking about “extract it from things that have been dead for 700,000 years” stable. If the instructions hung around in the cell for that long the factories would keep rereading them and producing waste disposal sites until the cell was one big scrapyard full of other, smaller scrapyards.
No, we need a message that is very “burn after reading”, and that’s where DNA’s older, more spontaneous cousin comes in, RNA.
RNA is, to put it lightly, temperamental. You blink and it’s gone, like butter in a hot pan. It needs to be stored at low temperatures to not immediately begin breaking down, and whilst this isn’t great for preserving a master copy of instructions (or for the patience of geneticists) it’s fantastic for short term messages you don’t want hanging around in your cell.

So these short term messages made of RNA, appropriately named “Messenger RNA” or mRNA for short, go out to the factories. The factories produce the building blocks, and the waste disposal site gets made.
“Fantastic!” I can hear you saying “but what’s this got to do with viruses?” That’s a good question. The answer is that this ‘message’ system has a fatal flaw. These factories will produce any messages that are sent to them. Even if the messages aren’t from our own cell.
Single (strand) and ready to mingle (with host DNA).
Our cells Part 2: Viral espionage
A virus is, in simplest terms, factory instructions wrapped in a protective packet. Coronaviruses are 70 – 150 nanometres across, which is ten millionth of a metre. (For context, a lung cell can be 5 million nanometres across, over 300,000 times larger).
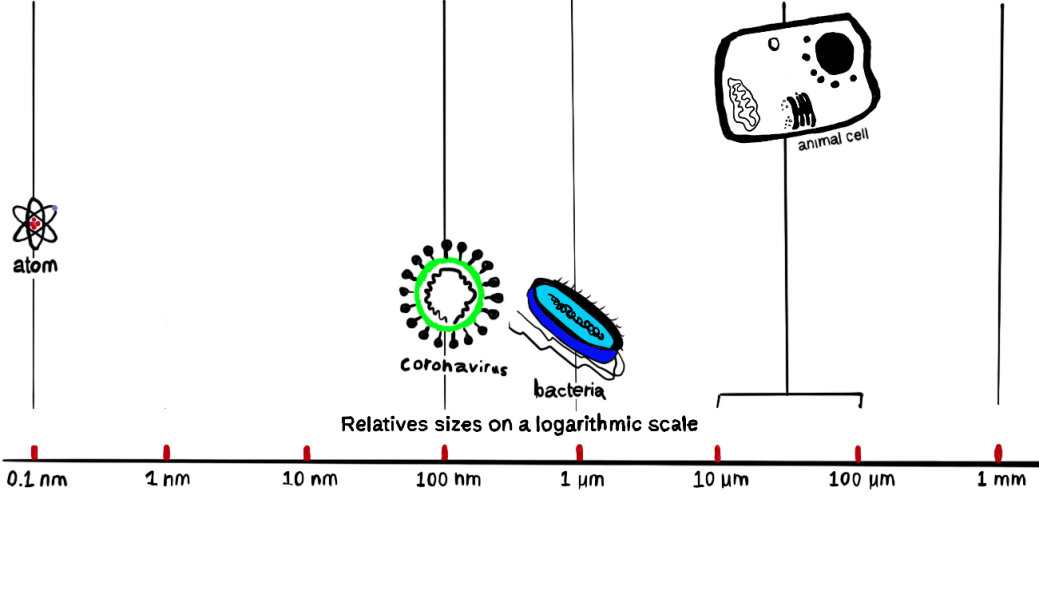
It’s sole job is to get into our cells, and make sure the instructions inside get read by our factories.
These instructions are in RNA, the same as our own, and whilst there are protective measures that destroy these viral instructions when they find them, if there’s enough virus some will find their way to the factories.
So what do these instructions say? Do they tell the cell to shut down? Do they take information from the cell?
No, the viral instructions do something even more impressive, they give cell instructions to make more of the virus itself. They repurpose the factory.

The virus hijacks the machinery of the cell, and since it doesn’t have to worry about maintaining a balance, it quickly overwhelms all other processes. It also has the ability to tear up your own messages so they don’t get read, giving more priority to its own instructions.
All these viral parts leave the factories and combine together into new viruses. The viral instructions overwhelm, until the cell is producing nothing but viral components and assembling them. The cell’s energy is being used up, it’s no longer transporting or sorting its own materials, and eventually, it bursts. The viruses within then move onto other cells to begin the process all over again.

This is the genius thing about viruses compared to bacteria and parasites, both of the latter are their own self-replicating organisms, which use their own processes to power themselves. They have their own factories and power plants. Viruses may seem lazy, but the line between laziness and efficiency, as I explain at work, is very small.
This seems ridiculous right? How can the cell not see through these fake instructions. How can it not see that they are damaging?
Cells have mechanisms to stop viruses, to identify and break up proteins, but if there are too many viruses already within the cell, they become overwhelmed. Viruses are small and quick to make, they don’t care for the safety of the cell, or for the larger effect their activity will have, all they want to do is replicate. They’ve hijacked your cell’s way of communicating. They put out false genetic statements that lead the cell to take the wrong actions. The message then leaves that cell and spreads to another, and another.
Imagine these viral instructions in your cells are words on your mobile phones, on your social media. Is the process so different? Isn’t it just the spreading of lies?
Conclusion: Of cells and cell phones
So what are the key points that link viruses with the system that spreads information about them?
Viruses and viral misinformation don’t need to be accurate.
Viruses and viral misinformation are shorter and don’t care about disrupting normal functioning.
Once there is too much virus or misinformation, the real information gets overwhelmed.
The COVID-19 virus has learned to lie, like the millions of viral ancestors before it. Viruses have perfected a form of genetic dishonesty that has plagued all living things, even bacteria, from the beginning of life on Earth. While we are fast developing tools to contest it, it has developed another layer of protection, and that is within the very hosts it’s trying to infect, spreading their own rumours and falsehoods. By saying the virus isn’t a threat, they’re mirroring the very thing they are denying.
The good news is the vast majority of viral infections end in defeat for the virus, our bodies have biological mechanisms to find and destroy the instructions, and cells can identify viral proteins within them and present these to the cell surface as “red flags”.
So if our cells can combat these viral misinformation so successfully, why do we as people still unknowingly spread online misinformation? Is there a mask to wear against this online onslaught? Are there red flags?
This is always a battle on two fronts, and while it may seem we can do little on the biological front, there is something you can do about the informational one. Whilst you’re social distancing, whilst you’re slowly meeting up with people again. Especially if we get a second wave…
Educate yourself. Educate others. This goes for this and any future situations like it. Don’t just believe what you read online and in the news. Check primary sources, and make sure any news articles you read cite the same sort of sources. Most importantly, always be thinking “Who is writing this? Is this person being honest with me, or are they trying to get me to repeat their instructions? Are they using me as a vector to pass on their dishonest advice so it will infect others?”
These are the viral words, meant to blend in with our real beliefs and replicate themselves. By recognizing this, we can fight the virus. We can see the misinformation instructions for what they are. And we can stop listening. Take a breath.
All of these posts on the crisis are drops in the ocean, as is mine. If you try to take them in all at once, it can feel like you’re drowning. So pause when you need to, float at the surface for a while. Because in this fight, the best treatment is a clear mind.

***
Real advice sources
In the UK, the best source for health info has always been the NHS website, or NICE (shameless self-promotion I know) for more in depth advice. It’s always from the primary source and made clear and easy to read.
In regards to wider health issues, whilst YouTube can just as easily host misinformation, there are great sources in the UK for clear unbiased information on COVID-19 and what you can do. These are not only about giving guidance, but about staying informed on the disease.
The government’s guidance isn’t always clearcut, and the best way to try and interpret it is arming yourself with knowledge of the virus and what’s happening.
Youtube:
- Dr John Campbell – Daily summary of virus figures and various discussions on treatment and government policy in the UK and around the world.
- Dr Hope’s Sick Notes – COVID-19 pandemic from a doctor’s perspective to give you an idea of how things are on the frontline
In general, look for articles that talk from a scientific perspective, they’ll be focusing on statistics, and they will not be talking in absolutes. It can be frustrating, but unfortunately these are unpredictable times, and its better to be honest when we don’t know something for sure, than to act like we’re certain.
For more on debunking coronavirus conspiracy theories, see the great Myles Power and his content, specifically his “capitalizing on corona” series.